Collagen vs. Collagen Peptides (What’s The Difference?)
Collagen is like the body’s construction crew—a protein composed of amino acids that hold the body’s structures together. It has benefits like improving joint health and soft tissue recovery, as well as improving skin health. However, when choosing to add a collagen supplement to your routine, which is best: collagen or collagen peptides?
Collagen peptides are collagen broken down into smaller “strands,” this breakdown process alters the functional properties and possibly the bio-availability (ability to absorb) of collagen. However, both collagen and collagen peptides can be beneficial and effective for health and recovery, and your stomach can also break down collagen into peptides in your stomach.
Most studies using collagen use collagen peptides, and it is generally recommended that you invest in collagen peptides above normal collagen.
Of course, context is king, so let’s dissect some of the literature behind collagen and collagen peptides and the differences between the two.
Understanding Collagen
Collagen is a fundamental protein in the human body, comprising approximately one-third of the total protein content. In contrast to whey protein, which significantly increases muscle mass, it plays a pivotal role in maintaining the mechanical properties of connective tissues and skin health [1].
Collagen is a key player in the extracellular matrix (ECM), which is like a crucial framework that helps keep tissues strong, regulated, and able to repair themselves [2].
Tendon health, integral to musculoskeletal function, is significantly influenced by collagen, constituting approximately 65-80% of its dry weight.
Collagen crosslinks within tendons are instrumental in fortifying structural integrity, enabling resilience against high-impact stresses and shear forces, particularly relevant in sports contexts [3].
Collagen has a unique structure, like a triple-helix, because of three important amino acids—glycine, proline, and hydroxyproline [4].
When collagen undergoes enzymatic hydrolysis, it turns into smaller bioactive peptides commonly found in collagen supplements.
These peptides are easily absorbed in the digestive tract before entering the bloodstream [5].
Collagen can come from different sources like bovine (cows), porcine (pigs), marine (fish), and poultry (chickens) [4].
What Are Collagen Peptides
First of all, what are peptides? Peptides are short chains of amino acids that serve as the fundamental building blocks of proteins.
Peptide bonds bind amino acids to give rise to peptides—almost like a string of beads. These peptides can then combine to build a protein.
Now, what happens if we flip this process? When collagen, a type of protein, is broken down, it transforms into peptides.
This breakdown occurs with the help of enzymes. These resulting collagen peptides have high absorbability.
Due to the enzyme-driven breakdown, collagen peptides differ from gelatin because they don’t create a gel-like consistency when absorbed in water, allowing them to completely dissolve.
Collagen vs. Collagen Peptides: Breaking Down The Differences
Composition And Structure
Collagen:
- Collagen is initially present in a full-length protein structure.
- This form is essentially the natural state of collagen within the body.
Collagen Peptides:
- Through enzymatic hydrolysis (breakdown), collagen transforms.
- When collagen undergoes hydrolysis, its structure changes from a triple-helix to a random coil due to the breaking of hydrogen bonds.
- This process results in the creation of collagen peptides, characterized by smaller, broken-down chains.
- Collagen peptides are distinct from the original full-length protein structure, making them more readily absorbable.
The image below shows how collagen is broken down into peptides (“native” collagen means that it is unchanged, “normal” collagen) and how their structures change throughout the process:
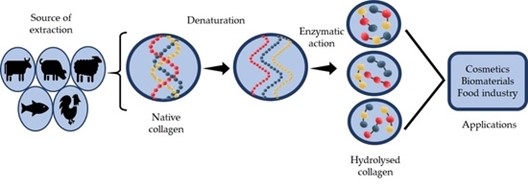
Figure 1: The breakdown of collagen [6]
Absorption And Bioavailability
Overall, collagen peptides may be more bioavailable than whole collagen because they are already broken down, but limited research is available on comparing collagen with collagen peptides.
Many studies, however, use collagen peptides rather than “normal collagen” to examine the impact of collagen on health.
Your stomach also contains enzymes that can break down collagen into peptides, so even if you consume collagen regularly, you will still reap its benefits.
Functional Properties
The composition and degree of hydrolysis (breakdown through enzymes) impact collagen’s functional properties, such as antioxidant capacity and antimicrobial activity.
These properties are mainly influenced by the molecular weight of the collagen [7, 8].
Collagen vs. Collagen Peptides Similarities
Both collagen and collagen peptides will yield the benefits of taking collagen, such as increased skin health, improved joint health, and soft tissue injury recovery—neither collagen nor collagen peptides form gels when dissolved in water.
Only gelatin, formed through heat treatment and the enzymatic breakdown of collagen, will form a gel when dissolved in water [9].
Both collagen and collagen peptides are tasteless unless they are found in a product with more ingredients.
Conclusion
When choosing between collagen and collagen peptides, both will benefit your health and performance in similar ways. However, if your budget allows, you can opt for collagen peptides, which can potentially be more bioavailable and have other functional benefits.
References
- Frantz, C., Stewart, K. M., & Weaver, V. M. (2010). The extracellular matrix at a glance. J Cell Sci, 123(Pt 24), 4195-4200. doi:10.1242/jcs.023820
- Iwai, K., Hasegawa, T., Taguchi, Y., Morimatsu, F., Sato, K., Nakamura, Y., . . . Ohtsuki, K. (2005). Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates. J Agric Food Chem, 53(16), 6531-6536. doi:10.1021/jf050206p
- Kjaer, M. (2004). Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiol Rev, 84(2), 649-698. doi:10.1152/physrev.00031.2003
- León-López, A., Morales-Peñaloza, A., Martínez-Juárez, V. M., Vargas-Torres, A., Zeugolis, D. I., & Aguirre-Álvarez, G. (2019). Hydrolyzed Collagen-Sources and Applications. Molecules, 24(22). doi:10.3390/molecules24224031
- León-López, A., Morales-Peñaloza, A., Martínez-Juárez, V. M., Vargas-Torres, A., Zeugolis, D. I., & Aguirre-Álvarez, G. (2019). Hydrolyzed Collagen—Sources and Applications. Molecules, 24(22), 4031. Retrieved from
- Li, Z., Wang, B., Chi, C., Gong, Y., Luo, H., & Ding, G. (2013). Influence of average molecular weight on antioxidant and functional properties of cartilage collagen hydrolysates from Sphyrna lewini, Dasyatis akjei and Raja porosa. Food Research International, 51(1), 283-293.
- Mokrejš, P., Gál, R., & Pavlačková, J. (2023). Enzyme Conditioning of Chicken Collagen and Taguchi Design of Experiments Enhancing the Yield and Quality of Prepared Gelatins. Int J Mol Sci, 24(4). doi:10.3390/ijms24043654
- Ricard-Blum, S. (2011). The collagen family. Cold Spring Harb Perspect Biol, 3(1), a004978. doi:10.1101/cshperspect.a004978
- Zhang, Y., Zhang, Y., Liu, X., Huang, L., Chen, Z., & Cheng, J. (2017). Influence of hydrolysis behaviour and microfluidisation on the functionality and structural properties of collagen hydrolysates. Food Chemistry, 227, 211-218.




